TGF-beta responding cancer stem cells: The culprit for metastasis and anti-cancer drug resistance
TGF-beta responding cancer stem cells: The culprit for metastasis and anti-cancer drug resistance
|
In general, tumors are an army of cancerous cells where they grow and divide excessively in the body. While we believed that tumor of the same origin possess the same physiological and biochemical characteristics, some cells succumb to certain anti-cancer drugs, while others may survive and bring the cancer back to the body when therapy ceases.
A group of scientists at Rockefeller University found the reason why cancer stem cells, the slow-growing tumor cells, were able to withstand anti-cancer therapy while the fast-dividing cancer cells couldn’t (Oshimori et al. 2015 Cell 160(5): 963-976). Their results point to the importance of the environment surrounding the cancer stem cells, specifically their exposure to the signal TGF-beta.
By visualizing TGF-beta signaling in mouse tumors using fluorescent reporter systems, the scientists found that cancer stem cells located nearest to the blood vessels receive the strongest TGF-beta signal. The TGF-beta responding stem cells proliferate more slowly but become invasive and metastatic over time. When the gene expression profiles of the slow-growing cancer stem cells was investigated, they found elevated expression of genes related to enzymes involved in anti-oxidant metabolism and detoxification in cells. Besides, TGF-beta transcriptionally activates p21 which stabilizes NRF2, thereby greatly enhance glutathione metabolism and increase drug resistance.
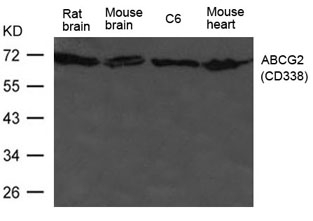
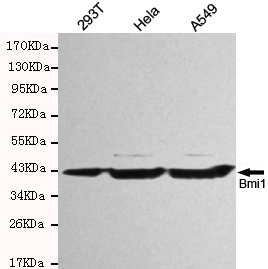
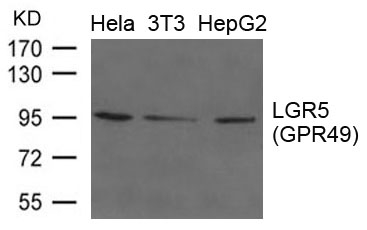
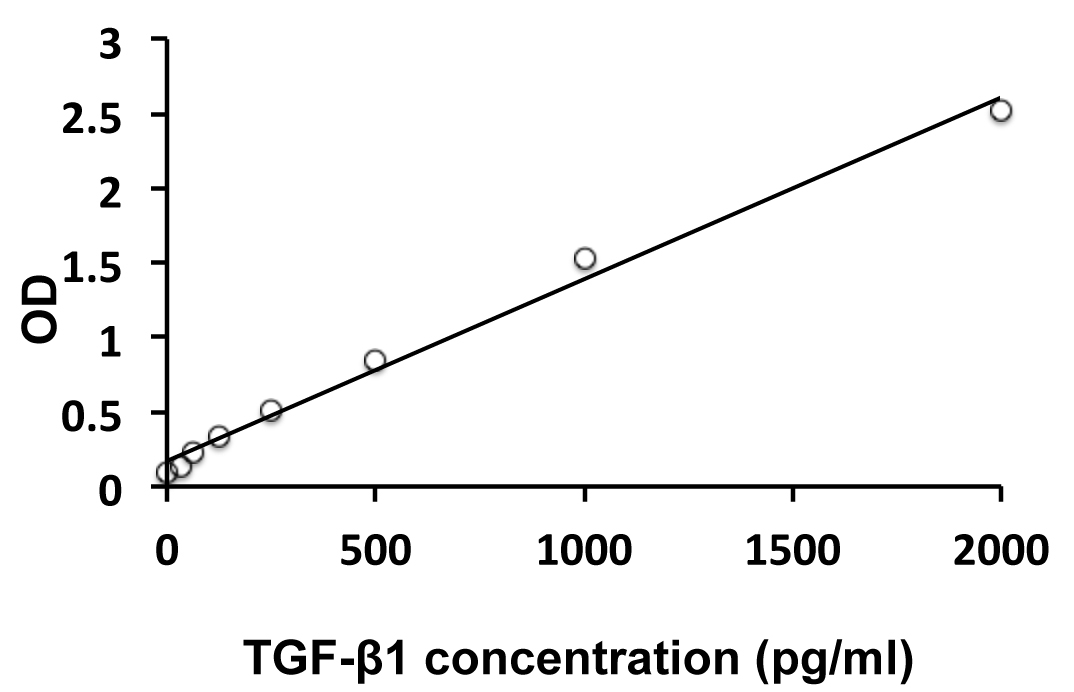
arigo’s Head and Neck Cancer Stem Cell Marker Panel (ARG30186) help to identify the slow-growing populations that might be resistant to anti-cancer drug. NRF-2 nuclear localization Antibody Duo (ARG30181) offers a good tool to study the nuclear-translocation and functions of NRF2 upon TGF-beta signaling. Furthermore, TGF-beta ELISA kits for mouse, human or rat are also available to quantify the level of TGF-beta in different tissue compartments (ARG80211, ARG80123 and ARG80245 respectively)
|
||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
For more product information, please contact Arigo Biolaboratories Corp. |
||||||||||||||||||||||||||||||
 |
||||||||||||||||||||||||||||||