|
 In the process of eliminating cancer cells or chronic viral infections, T cell exhaustion might occur due to the overexpression of coinhibitory receptors such as PD-1 and CTLA-4, rendering T cell inactive and failure to combat diseases. Many cancer therapies were developed based on the principle of blocking coinhibitory molecule on the surface of exhausted T cells. However, partial clinical responses on patients suggest that more receptors might be responsible in the induction of T cell exhaustion, suggesting that combined therapeutic programs might be necessary. In the process of eliminating cancer cells or chronic viral infections, T cell exhaustion might occur due to the overexpression of coinhibitory receptors such as PD-1 and CTLA-4, rendering T cell inactive and failure to combat diseases. Many cancer therapies were developed based on the principle of blocking coinhibitory molecule on the surface of exhausted T cells. However, partial clinical responses on patients suggest that more receptors might be responsible in the induction of T cell exhaustion, suggesting that combined therapeutic programs might be necessary.
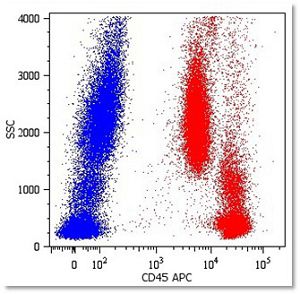
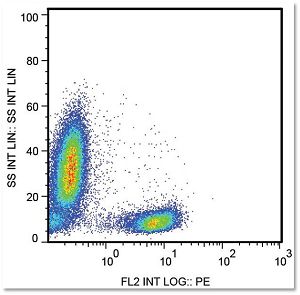
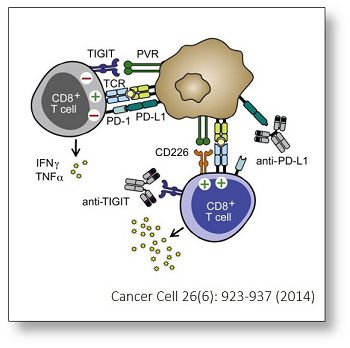
Johnston RJ et al. found that blocking TIGIT (T cell immunoglobulin and ITIM domain) in mice receiving anti-PD1 therapy improved effector activity of CD8+ T cells through the secretion of IFN-gamma and TNF-alpha, resulting in significant tumor and viral clearance. [Johnston RJ et al. (2014). Cancer Cell 26(6): 923-937]. They also showed that TIGIT is highly upregulated on tumor-infiltrating T cells, and functionally blocking CD226 (a competitor of TIGIT for PVR ligand) counteracted the combinatorial therapy-induced tumor regression in mice. This research unveils the potential of PD-1 and TIGIT combined therapy in improving the immune responses of patients with cancer or chronic viral infections.
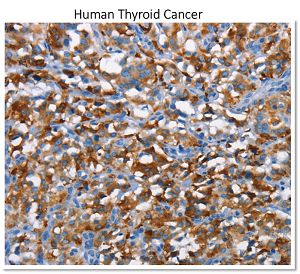
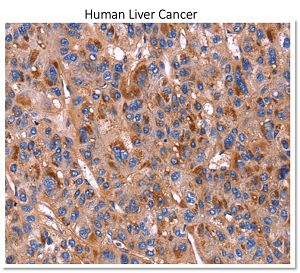
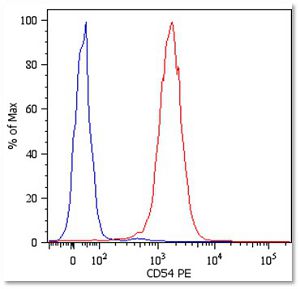
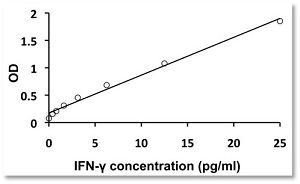
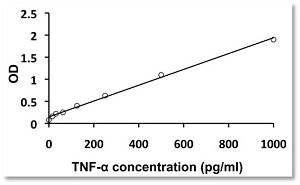
In arigo, we provide T Cell Intercellular Adhesion Molecule Duo (CD226, PVR) (ARG30163) to investigate the competitive roles of TIGIT and CD226 for PVR ligand. Cytotoxic T Cell Surface Marker Antibody Panel (FACS) (ARG30162) assists researchers to identify and isolate this subset of T cells responsible for combating tumor or viral infections. IFN gamma ELISA Kit (ARG80116) and TNF alpha ELISA Kit (ARG80120) allow researchers to evaluate the effector activity of CD8+ T cells upon drug treatments.
|



 In the process of eliminating cancer cells or chronic viral infections, T cell exhaustion might occur due to the overexpression of coinhibitory receptors such as PD-1 and CTLA-4, rendering T cell inactive and failure to combat diseases. Many cancer therapies were developed based on the principle of blocking coinhibitory molecule on the surface of exhausted T cells. However, partial clinical responses on patients suggest that more receptors might be responsible in the induction of T cell exhaustion, suggesting that combined therapeutic programs might be necessary.
In the process of eliminating cancer cells or chronic viral infections, T cell exhaustion might occur due to the overexpression of coinhibitory receptors such as PD-1 and CTLA-4, rendering T cell inactive and failure to combat diseases. Many cancer therapies were developed based on the principle of blocking coinhibitory molecule on the surface of exhausted T cells. However, partial clinical responses on patients suggest that more receptors might be responsible in the induction of T cell exhaustion, suggesting that combined therapeutic programs might be necessary.