ARG10314
anti-Influenza A nucleoprotein antibody [F8]
anti-Influenza A nucleoprotein antibody [F8] for ELISA,ICC/IF and Influenza A virus
Microbiology and Infectious Disease antibody
Overview
| Product Description | Mouse Monoclonal antibody [F8] recognizes Influenza A nucleoprotein |
|---|---|
| Tested Reactivity | Influenza A |
| Tested Application | ELISA, ICC/IF |
| Specificity | No cross reactivity to NP of Influenza B virus. |
| Host | Mouse |
| Clonality | Monoclonal |
| Clone | F8 |
| Isotype | IgG2a |
| Target Name | Influenza A nucleoprotein |
| Immunogen | purified influenza virus type A strain H1N1 |
| Conjugation | Un-conjugated |
Application Instructions
| Application Note | * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
|---|
Properties
| Form | Liquid |
|---|---|
| Purification | Protein G affinity purified. |
| Buffer | PBS (pH 7.4) and 0.1% Sodium azide |
| Preservative | 0.1% Sodium azide |
| Concentration | 1.0-2.0 mg/ml |
| Storage Instruction | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| Note | For laboratory research only, not for drug, diagnostic or other use. |
Bioinformation
| Database Links | |
|---|---|
| Gene Symbol | NP |
| Gene Full Name | nucleoprotein |
| Research Area | Microbiology and Infectious Disease antibody |
Images (1) Click the Picture to Zoom In
-
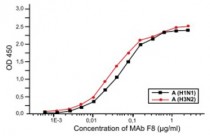
ARG10314 anti-Influenza A nucleoprotein antibody [F8] ELISA image
ELISA: Specific activity of MAb F8 in ELISA with purified virus antigens A (H1N1) and A (H3N2). The investigation of F8 MAb specificity showed that it recognizes the conservative epitope expressed on the nucleoprotein, which is common for type A viruses with different antigenic structure and species origin. We investigated 25 strains of human and avian influenza virus A, isolated during different epidemics in the period from 1934 till 1993 and in all the cases specific reaction was observed. We investigated 265 samples of nasal washings from patients during influenza outbreaks in children’s communities by the method of direct immunofluorescense. Sensitivity and specificity of the influenza virus A detection reached 60 % and 98.2 % respectively.