ARG66647
anti-Myelin Basic Protein antibody [SQab19166]
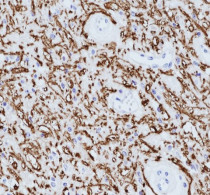
anti-Myelin Basic Protein antibody [SQab19166] for IHC-Formalin-fixed paraffin-embedded sections and Human
Overview
| Product Description | Recombinant Rabbit Monoclonal antibody [SQab19166] recognizes Myelin Basic Protein |
|---|---|
| Tested Reactivity | Hu |
| Tested Application | IHC-P |
| Host | Rabbit |
| Clonality | Monoclonal |
| Clone | SQab19166 |
| Isotype | IgG |
| Target Name | Myelin Basic Protein |
| Antigen Species | Human |
| Immunogen | Synthetic peptide within aa. 150-250 of Human Myelin Basic Protein. |
| Conjugation | Un-conjugated |
| Alternate Names | Myelin A1 protein; MBP; Myelin membrane encephalitogenic protein; Myelin basic protein |
Application Instructions
| Application Suggestion |
|
||||
|---|---|---|---|---|---|
| Application Note | IHC-P: Antigen Retrieval: Heat mediation was performed in Tris/EDTA buffer (pH 9.0), primary antibody incubate at RT (18°C-25°C) for 30 minutes. * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
Properties
| Form | Liquid |
|---|---|
| Purification | Purification with Protein A. |
| Buffer | PBS, 0.01% Sodium azide, 40% Glycerol and 0.05% BSA. |
| Preservative | 0.01% Sodium azide |
| Stabilizer | 40% Glycerol and 0.05% BSA |
| Storage Instruction | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| Note | For laboratory research only, not for drug, diagnostic or other use. |
Bioinformation
| Database Links | |
|---|---|
| Gene Symbol | MBP |
| Gene Full Name | myelin basic protein |
| Background | The protein encoded by the classic MBP gene is a major constituent of the myelin sheath of oligodendrocytes and Schwann cells in the nervous system. However, MBP-related transcripts are also present in the bone marrow and the immune system. These mRNAs arise from the long MBP gene (otherwise called "Golli-MBP") that contains 3 additional exons located upstream of the classic MBP exons. Alternative splicing from the Golli and the MBP transcription start sites gives rise to 2 sets of MBP-related transcripts and gene products. The Golli mRNAs contain 3 exons unique to Golli-MBP, spliced in-frame to 1 or more MBP exons. They encode hybrid proteins that have N-terminal Golli aa sequence linked to MBP aa sequence. The second family of transcripts contain only MBP exons and produce the well characterized myelin basic proteins. This complex gene structure is conserved among species suggesting that the MBP transcription unit is an integral part of the Golli transcription unit and that this arrangement is important for the function and/or regulation of these genes. [provided by RefSeq, Jul 2008] |
| Function | The classic group of MBP isoforms (isoform 4-isoform 14) are with PLP the most abundant protein components of the myelin membrane in the CNS. They have a role in both its formation and stabilization. The smaller isoforms might have an important role in remyelination of denuded axons in multiple sclerosis. The non-classic group of MBP isoforms (isoform 1-isoform 3/Golli-MBPs) may preferentially have a role in the early developing brain long before myelination, maybe as components of transcriptional complexes, and may also be involved in signaling pathways in T-cells and neural cells. Differential splicing events combined with optional post-translational modifications give a wide spectrum of isomers, with each of them potentially having a specialized function. Induces T-cell proliferation. [UniProt] |
| Cellular Localization | Myelin membrane; Peripheral membrane protein; Cytoplasmic side. Note=Cytoplasmic side of myelin. Isoform 3: Nucleus. Note=Targeted to nucleus in oligodendrocytes. [UniProt] |
| Calculated MW | 33 kDa |
| PTM | Several charge isomers of MBP; C1 (the most cationic, least modified, and most abundant form), C2, C3, C4, C5, C6, C7, C8-A and C8-B (the least cationic form); are produced as a result of optional PTM, such as phosphorylation, deamidation of glutamine or asparagine, arginine citrullination and methylation. C8-A and C8-B contain each two mass isoforms termed C8-A(H), C8-A(L), C8-B(H) and C8-B(L), (H) standing for higher and (L) for lower molecular weight. C3, C4 and C5 are phosphorylated. The ratio of methylated arginine residues decreases during aging, making the protein more cationic. The N-terminal alanine is acetylated (isoform 3, isoform 4, isoform 5 and isoform 6). Arg-241 was found to be 6% monomethylated and 60% symmetrically dimethylated. Phosphorylated by TAOK2, VRK2, MAPK11, MAPK12, MAPK14 and MINK1. [UniProt] |
Images (1) Click the Picture to Zoom In