ARG22294
anti-SOD1 antibody
anti-SOD1 antibody for ELISA,IHC-Formalin-fixed paraffin-embedded sections,Immunoprecipitation,Western blot and Bovine,Dog,Fish,Hamster,Human,Insect,Invertebrates,Monkey,Mouse,Pig,Rabbit,Rat,Sheep,Xenopus laevis
Overview
| Product Description | Rabbit Polyclonal antibody recognizes SOD1 |
|---|---|
| Tested Reactivity | Hu, Ms, Rat, Bov, Dog, Fsh, Hm, Insect, Invt, Mk, Pig, Rb, Sheep, Xenopus laevis |
| Tested Application | ELISA, IHC-P, IP, WB |
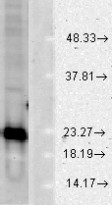
| Specificity | Detects ~23kDa (human) and ~19kDa (other species). |
| Host | Rabbit |
| Clonality | Polyclonal |
| Target Name | SOD1 |
| Antigen Species | Human |
| Immunogen | Human SOD1 |
| Conjugation | Un-conjugated |
| Alternate Names | homodimer; EC 1.15.1.1; SOD; HEL-S-44; Superoxide dismutase [Cu-Zn]; ALS1; Superoxide dismutase 1; IPOA; ALS; hSod1 |
Application Instructions
| Application Suggestion |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Application Note | * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
Properties
| Form | Liquid |
|---|---|
| Purification | Purification with Protein A. |
| Buffer | PBS (pH 7.0), 0.09% Sodium azide and 50% Glycerol |
| Preservative | 0.09% Sodium azide |
| Stabilizer | 50% Glycerol |
| Concentration | 1 mg/ml |
| Storage Instruction | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| Note | For laboratory research only, not for drug, diagnostic or other use. |
Bioinformation
| Database Links | |
|---|---|
| Gene Symbol | SOD1 |
| Gene Full Name | superoxide dismutase 1, soluble |
| Background | The protein encoded by this gene binds copper and zinc ions and is one of two isozymes responsible for destroying free superoxide radicals in the body. The encoded isozyme is a soluble cytoplasmic protein, acting as a homodimer to convert naturally-occuring but harmful superoxide radicals to molecular oxygen and hydrogen peroxide. The other isozyme is a mitochondrial protein. Mutations in this gene have been implicated as causes of familial amyotrophic lateral sclerosis. Rare transcript variants have been reported for this gene. [provided by RefSeq, Jul 2008] |
| Function | Destroys radicals which are normally produced within the cells and which are toxic to biological systems. [UniProt] |
| Cellular Localization | Cytoplasm |
| Calculated MW | ~23 kDa (human) and ~19 kDa (other species). |
| PTM | Unlike wild-type protein, the pathogenic variants ALS1 Arg-38, Arg-47, Arg-86 and Ala-94 are polyubiquitinated by RNF19A leading to their proteasomal degradation. The pathogenic variants ALS1 Arg-86 and Ala-94 are ubiquitinated by MARCH5 leading to their proteasomal degradation. The ditryptophan cross-link at Trp-33 is responsible for the non-disulfide-linked homodimerization. Such modification might only occur in extreme conditions and additional experimental evidence is required. Palmitoylation helps nuclear targeting and decreases catalytic activity. Succinylation, adjacent to copper catalytic site, probably inhibits activity. Desuccinylation by SIRT5 enhances activity. |
Images (2) Click the Picture to Zoom In
-
ARG22294 anti-SOD1 antibody WB image
Western blot: Mouse hippocampus stained with ARG22294 anti-SOD1 antibody, ARG54937 anti-SOD2 antibody and ARG65683 anti-beta Actin antibody.
From Zihao Xia et al. International Journal o f Molecular Sciences (2022), doi: 10.3390/ijms23126463, Fig. 6C.
-
ARG22294 anti-Cu/Zn-SOD antibody WB image
Western blot: Human cell line lysates stained with ARG22294 anti-Cu/Zn-SOD antibody at 1:1000 dilution.