ARG20573
anti-c Abl phospho (Tyr412) antibody
anti-c Abl phospho (Tyr412) antibody for Western blot and Human
Cancer antibody; Gene Regulation antibody; Signaling Transduction antibody
Overview
| Product Description | Rabbit Polyclonal antibody recognizes c Abl phospho (Tyr412) |
|---|---|
| Tested Reactivity | Hu |
| Predict Reactivity | Ms, Rat |
| Tested Application | WB |
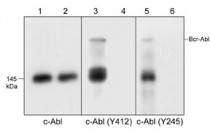
| Specificity | This antibody was cross-adsorbed to phospho-tyrosine coupled to agarose and to phospho-c-Abl (Tyr-245) peptide before affinity purification using phospho-c-Abl (Tyr-412) peptide. On SDS-PAGE immunoblots of K-562 treated with pervanadate the antibody detects a ~145 kDa protein corresponding to c-Abl and a 210 kDa band corresponding to BCR-Abl. |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | IgG |
| Target Name | c Abl |
| Antigen Species | Human |
| Immunogen | KLH-conjugated phosphospecific peptide around Tyr412 of Human c-Abl. This peptide sequence has high homology to the conserved site in Rat and Mouse c-Abl, as well as in viral Abl and BCR-Abl fusion protein. |
| Conjugation | Un-conjugated |
| Alternate Names | bcr/abl; Proto-oncogene c-Abl; c-ABL; Abelson murine leukemia viral oncogene homolog 1; ABL; Abelson tyrosine-protein kinase 1; JTK7; Tyrosine-protein kinase ABL1; p150; v-abl; c-ABL1; EC 2.7.10.2 |
Application Instructions
| Application Suggestion |
|
||||
|---|---|---|---|---|---|
| Application Note | WB: Antibody is suggested to be diluted in 5% skimmed milk/Tris buffer with 0.04% Tween20 and incubated for 1 hour at room temperature. * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
Properties
| Form | Liquid |
|---|---|
| Purification | Affinity purification with immunogen. |
| Buffer | 100 μl PBS, 50% Glycerol, 1 mg/ml BSA and 0.05% Sodium azide |
| Preservative | 0.05% Sodium azide |
| Stabilizer | 50% Glycerol, 1 mg/ml BSA |
| Storage Instruction | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| Note | For laboratory research only, not for drug, diagnostic or other use. |
Bioinformation
| Database Links | |
|---|---|
| Gene Symbol | ABL1 |
| Gene Full Name | ABL proto-oncogene 1, non-receptor tyrosine kinase |
| Background | This gene is a protooncogene that encodes a protein tyrosine kinase involved in a variety of cellular processes, including cell division, adhesion, differentiation, and response to stress. The activity of the protein is negatively regulated by its SH3 domain, whereby deletion of the region encoding this domain results in an oncogene. The ubiquitously expressed protein has DNA-binding activity that is regulated by CDC2-mediated phosphorylation, suggesting a cell cycle function. This gene has been found fused to a variety of translocation partner genes in various leukemias, most notably the t(9;22) translocation that results in a fusion with the 5' end of the breakpoint cluster region gene (BCR; MIM:151410). Alternative splicing of this gene results in two transcript variants, which contain alternative first exons that are spliced to the remaining common exons. [provided by RefSeq, Aug 2014] |
| Function | Non-receptor tyrosine-protein kinase that plays a role in many key processes linked to cell growth and survival such as cytoskeleton remodeling in response to extracellular stimuli, cell motility and adhesion, receptor endocytosis, autophagy, DNA damage response and apoptosis. Coordinates actin remodeling through tyrosine phosphorylation of proteins controlling cytoskeleton dynamics like WASF3 (involved in branch formation); ANXA1 (involved in membrane anchoring); DBN1, DBNL, CTTN, RAPH1 and ENAH (involved in signaling); or MAPT and PXN (microtubule-binding proteins). Phosphorylation of WASF3 is critical for the stimulation of lamellipodia formation and cell migration. Involved in the regulation of cell adhesion and motility through phosphorylation of key regulators of these processes such as BCAR1, CRK, CRKL, DOK1, EFS or NEDD9. Phosphorylates multiple receptor tyrosine kinases and more particularly promotes endocytosis of EGFR, facilitates the formation of neuromuscular synapses through MUSK, inhibits PDGFRB-mediated chemotaxis and modulates the endocytosis of activated B-cell receptor complexes. Other substrates which are involved in endocytosis regulation are the caveolin (CAV1) and RIN1. Moreover, ABL1 regulates the CBL family of ubiquitin ligases that drive receptor down-regulation and actin remodeling. Phosphorylation of CBL leads to increased EGFR stability. Involved in late-stage autophagy by regulating positively the trafficking and function of lysosomal components. ABL1 targets to mitochondria in response to oxidative stress and thereby mediates mitochondrial dysfunction and cell death. ABL1 is also translocated in the nucleus where it has DNA-binding activity and is involved in DNA-damage response and apoptosis. Many substrates are known mediators of DNA repair: DDB1, DDB2, ERCC3, ERCC6, RAD9A, RAD51, RAD52 or WRN. Activates the proapoptotic pathway when the DNA damage is too severe to be repaired. Phosphorylates TP73, a primary regulator for this type of damage-induced apoptosis. Phosphorylates the caspase CASP9 on 'Tyr-153' and regulates its processing in the apoptotic response to DNA damage. Phosphorylates PSMA7 that leads to an inhibition of proteasomal activity and cell cycle transition blocks. ABL1 acts also as a regulator of multiple pathological signaling cascades during infection. Several known tyrosine-phosphorylated microbial proteins have been identified as ABL1 substrates. This is the case of A36R of Vaccinia virus, Tir (translocated intimin receptor) of pathogenic E.coli and possibly Citrobacter, CagA (cytotoxin-associated gene A) of H.pylori, or AnkA (ankyrin repeat-containing protein A) of A.phagocytophilum. Pathogens can highjack ABL1 kinase signaling to reorganize the host actin cytoskeleton for multiple purposes, like facilitating intracellular movement and host cell exit. Finally, functions as its own regulator through autocatalytic activity as well as through phosphorylation of its inhibitor, ABI1. [UniProt] |
| Research Area | Cancer antibody; Gene Regulation antibody; Signaling Transduction antibody |
| Calculated MW | 123 kDa |
| PTM | Acetylated at Lys-711 by EP300 which promotes the cytoplasmic translocation. Phosphorylation at Tyr-70 by members of the SRC family of kinases disrupts SH3 domain-based autoinhibitory interactions and intermolecular associations, such as that with ABI1, and also enhances kinase activity. Phosphorylation at Tyr-226 and Tyr-393 correlate with increased activity. DNA damage-induced activation of ABL1 requires the function of ATM and Ser-446 phosphorylation (By similarity). Phosphorylation at Ser-569 has been attributed to a CDC2-associated kinase and is coupled to cell division (By similarity). Phosphorylation at Ser-618 and Ser-619 by PAK2 increases binding to CRK and reduces binding to ABI1. Phosphorylation on Thr-735 is required for binding 14-3-3 proteins for cytoplasmic translocation. Phosphorylated by PRKDC (By similarity). Polyubiquitinated. Polyubiquitination of ABL1 leads to degradation. |
Images (1) Click the Picture to Zoom In
-
ARG20573 anti-c Abl phospho (Tyr412) antibody WB image
Western blot: K-562 cells treated with pervanadate (1 mM) for 30 minutes (Lanes 1, 3, 5) or treated with alkaline phosphatase to remove phosphorylation on c-Abl (Lanes 2, 4, 6), then the blots were stained with ARG20570 anti-c-Abl antibody [M209] (Lanes 1, 2), ARG20573 anti-c Abl phospho (Tyr412) antibody (Lanes 3, 4), or ARG20569 anti-c-Abl phospho (Tyr245) antibody (Lanes 5, 6).