ARG10515
anti-HIF-1 alpha antibody [ESEE122]
anti-HIF-1 alpha antibody [ESEE122] for ELISA,ICC/IF,IHC-Formalin-fixed paraffin-embedded sections,IHC-Frozen sections,Western blot and Human,Mouse,Rat
Cancer antibody; Cell Biology and Cellular Response antibody; Gene Regulation antibody; Metabolism antibody

Overview
| Product Description | Mouse Monoclonal antibody [ESEE122] recognizes HIF-1 alpha |
|---|---|
| Tested Reactivity | Hu, Ms, Rat |
| Tested Application | ELISA, ICC/IF, IHC-Fr, IHC-P, WB |
| Host | Mouse |
| Clonality | Monoclonal |
| Clone | ESEE122 |
| Isotype | IgG1 |
| Target Name | HIF-1 alpha |
| Antigen Species | Human |
| Immunogen | Synthetic peptide within 329 to 530 aa of Human HIF-1A protein (NP_001230013.1) |
| Conjugation | Un-conjugated |
| Alternate Names | Class E basic helix-loop-helix protein 78; Basic-helix-loop-helix-PAS protein MOP1; Hypoxia-inducible factor 1-alpha; PAS domain-containing protein 8; HIF1-alpha; HIF1-ALPHA; HIF1; MOP1; HIF-1-alpha; PASD8; HIF-1A; HIF-1alpha; Member of PAS protein 1; ARNT-interacting protein; bHLHe78 |
Application Instructions
| Application Suggestion |
|
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Application Note | IHC-P: Antigen Retrieval: By heat mediation. * The dilutions indicate recommended starting dilutions and the optimal dilutions or concentrations should be determined by the scientist. |
||||||||||||
| Positive Control | Hypoxic cell pellets. | ||||||||||||
| Observed Size | ~130 kDa |
Properties
| Form | Liquid |
|---|---|
| Purification | Purified by affinity chromatography. |
| Buffer | PBS and 0.02% Sodium azide |
| Preservative | 0.02% Sodium azide |
| Concentration | 1 mg/ml |
| Storage Instruction | For continuous use, store undiluted antibody at 2-8°C for up to a week. For long-term storage, aliquot and store at -20°C or below. Storage in frost free freezers is not recommended. Avoid repeated freeze/thaw cycles. Suggest spin the vial prior to opening. The antibody solution should be gently mixed before use. |
| Note | For laboratory research only, not for drug, diagnostic or other use. |
Bioinformation
| Database Links | |
|---|---|
| Gene Symbol | HIF1A |
| Gene Full Name | hypoxia inducible factor 1, alpha subunit (basic helix-loop-helix transcription factor) |
| Background | The alpha subunit of transcription factor hypoxia-inducible factor-1 (HIF-1), which is a heterodimer composed of an alpha and a beta subunit. HIF-1 functions as a master regulator of cellular and systemic homeostatic response to hypoxia by activating transcription of many genes, including those involved in energy metabolism, angiogenesis, apoptosis, and other genes whose protein products increase oxygen delivery or facilitate metabolic adaptation to hypoxia. HIF-1 thus plays an essential role in embryonic vascularization, tumor angiogenesis and pathophysiology of ischemic disease. Alternatively spliced transcript variants encoding different isoforms have been identified for this gene. [provided by RefSeq, Jul 2011] |
| Function | Functions as a master transcriptional regulator of the adaptive response to hypoxia. Under hypoxic conditions, activates the transcription of over 40 genes, including erythropoietin, glucose transporters, glycolytic enzymes, vascular endothelial growth factor, HILPDA, and other genes whose protein products increase oxygen delivery or facilitate metabolic adaptation to hypoxia. Plays an essential role in embryonic vascularization, tumor angiogenesis and pathophysiology of ischemic disease. Binds to core DNA sequence 5'-[AG]CGTG-3' within the hypoxia response element (HRE) of target gene promoters. Activation requires recruitment of transcriptional coactivators such as CREBPB and EP300. Activity is enhanced by interaction with both, NCOA1 or NCOA2. Interaction with redox regulatory protein APEX seems to activate CTAD and potentiates activation by NCOA1 and CREBBP. Involved in the axonal distribution and transport of mitochondria in neurons during hypoxia. [Uniprot] |
| Highlight | Related products: HIF1 alpha antibodies; HIF1 alpha ELISA Kits; Anti-Mouse IgG secondary antibodies; Related news: Baking soda restores circadian clock in tumor cells Hypoxia-induced transcription, histone demethylases are involved |
| Research Area | Cancer antibody; Cell Biology and Cellular Response antibody; Gene Regulation antibody; Metabolism antibody |
| Calculated MW | 93 kDa |
| PTM | In normoxia, is hydroxylated on Pro-402 and Pro-564 in the oxygen-dependent degradation domain (ODD) by EGLN1/PHD2 and EGLN2/PHD1. EGLN3/PHD3 has also been shown to hydroxylate Pro-564. The hydroxylated prolines promote interaction with VHL, initiating rapid ubiquitination and subsequent proteasomal degradation. Deubiquitinated by USP20. Under hypoxia, proline hydroxylation is impaired and ubiquitination is attenuated, resulting in stabilization. In normoxia, is hydroxylated on Asn-803 by HIF1AN, thus abrogating interaction with CREBBP and EP300 and preventing transcriptional activation. This hydroxylation is inhibited by the Cu/Zn-chelator, Clioquinol. S-nitrosylation of Cys-800 may be responsible for increased recruitment of p300 coactivator necessary for transcriptional activity of HIF-1 complex. Requires phosphorylation for DNA-binding. Phosphorylation at Ser-247 by CSNK1D/CK1 represses kinase activity and impairs ARNT binding. Phosphorylation by GSK3-beta and PLK3 promote degradation by the proteasome. Sumoylated; with SUMO1 under hypoxia. Sumoylation is enhanced through interaction with RWDD3. Both sumoylation and desumoylation seem to be involved in the regulation of its stability during hypoxia. Sumoylation can promote either its stabilization or its VHL-dependent degradation by promoting hydroxyproline-independent HIF1A-VHL complex binding, thus leading to HIF1A ubiquitination and proteasomal degradation. Desumoylation by SENP1 increases its stability amd transcriptional activity. There is a disaccord between various publications on the effect of sumoylation and desumoylation on its stability and transcriptional activity. Acetylation of Lys-532 by ARD1 increases interaction with VHL and stimulates subsequent proteasomal degradation (PubMed:12464182). Deacetylation of Lys-709 by SIRT2 increases its interaction with and hydroxylation by EGLN1 thereby inactivating HIF1A activity by inducing its proteasomal degradation (PubMed:24681946). Polyubiquitinated; in normoxia, following hydroxylation and interaction with VHL. Lys-532 appears to be the principal site of ubiquitination. Clioquinol, the Cu/Zn-chelator, inhibits ubiquitination through preventing hydroxylation at Asn-803. Ubiquitinated by a CUL2-based E3 ligase. The iron and 2-oxoglutarate dependent 3-hydroxylation of asparagine is (S) stereospecific within HIF CTAD domains. |
Images (3) Click the Picture to Zoom In
-
ARG10515 anti-HIF-1 alpha antibody [ESEE122] IHC-P image
Immunohistochemistry: Paraffin-embedded Mouse muscle tissue stained with ARG10515 anti-HIF-1 alpha antibody [ESEE122] at 1:90 dilution for 2 hours at 37°C.
-
ARG10515 anti-HIF-1 alpha [ESEE122] antibody WB image
Western blot: Whole cell extracts from HeLa (normoxia) or HeLa (0.1% hypoxia for 4 hours) stained with ARG10515 anti-HIF-1 alpha [ESEE122] antibody at 4 μg/ml dilution. (Clone WB data from Am J Pathol. 2000 Aug; 157(2): 411–421; PMID: 10934146)
-
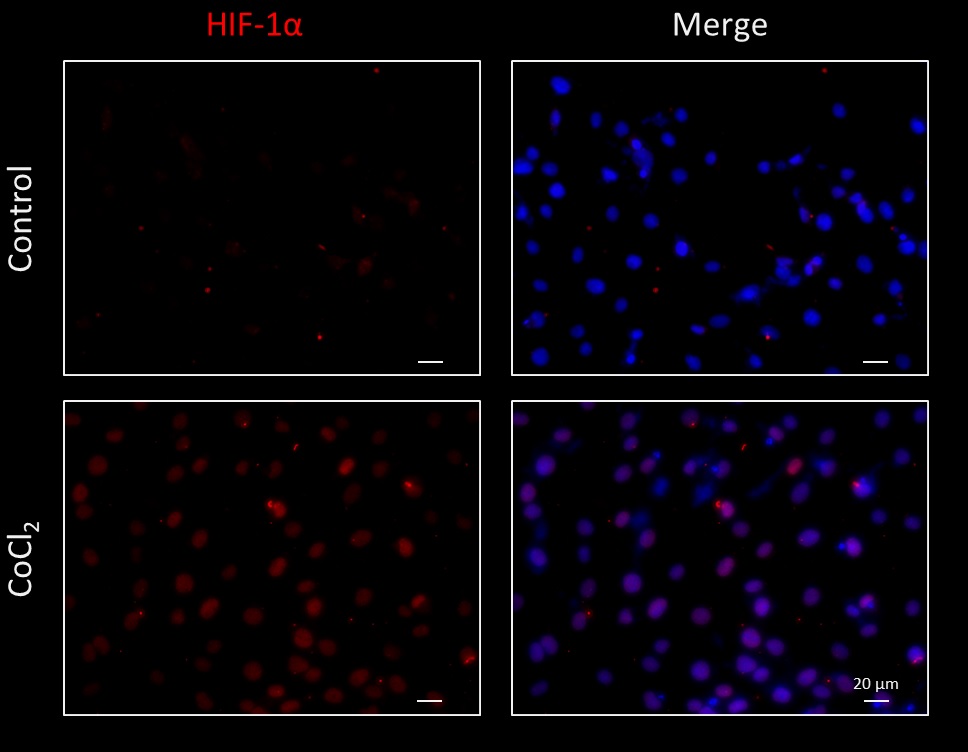
ARG10515 anti-HIF-1 alpha antibody [ESEE122] ICC/IF image
Immunofluorescence: NRK-49F cells untreated or treated with CoCl2 (400 μM) for 8 hours. Cells were fixed with 4% PFA for 15 min at room temperature and permeabilized by 0.5% Triton X-100 for 10 min at room temperature. Blocking: CAS-Blocking buffer for 1 hour. Cells were stained with ARG10515 anti-HIF-1 alpha antibody [ESEE122] (red) at 1:200 dilution, overnight at 4°C, in filtered PBS. DAPI (blue) for nuclear staining.
Customer's Feedback
 Excellent
Excellent
Review for anti-HIF-1 alpha antibody [ESEE122]
Application:IF/ICC
Sample:NRK-49F untreated or treated with CoCl2 (400 μM) for 8 h
Fixation Buffer:4% PFA
Fixation Time:15 min
Fixation Temperature:20 - 25 ºC
Permeabilization Buffer:0.5% Triton X-100
Primary Antibody Dilution Factor:1:200
Primary Antibody Incubation Time:overnight
Primary Antibody Incubation Temperature:4 ºC
Conjugation of Secondary Antibody:Alexa Fluor 594
Specific References